The NA-ACCORD welcomes all who are interested to participate in research.
- 1. Getting started
Have a research question? Start by reading the NA-ACCORD’s Collaboration Polices and an overview of the steps required in our proposal development process. Then, complete the NA-ACCORD Concept Sheet to submit your idea for approval. You may find our list of Available Data Elements helpful in drafting your Concept Sheet.
- 2. Concept Sheet approval process
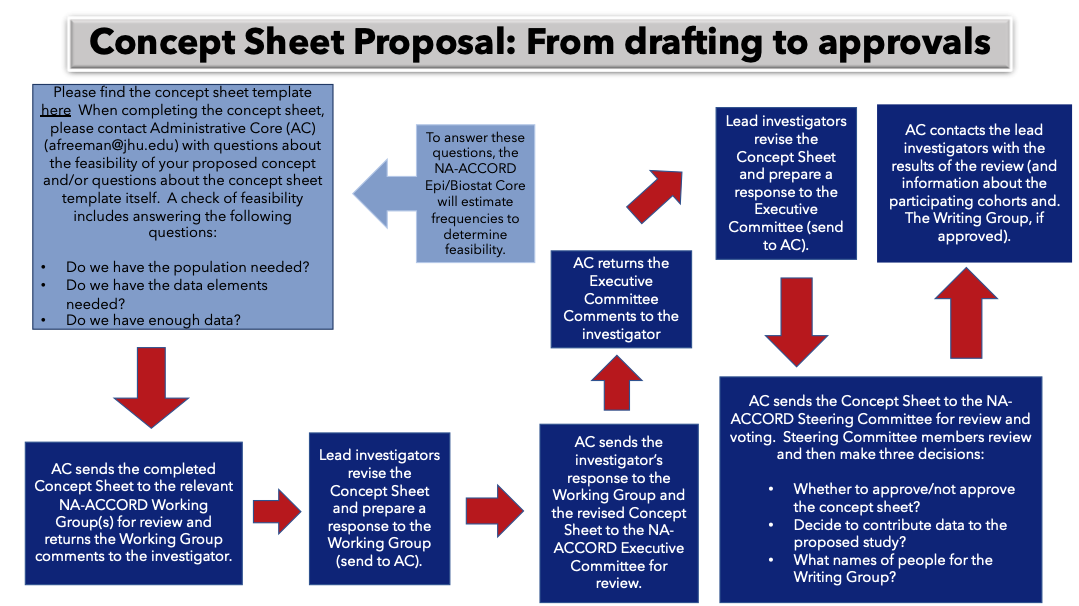
Your Concept Sheet will be reviewed by:
- The appropriate NA-ACCORD Scientific Working Group
- The NA-ACCORD Executive Committee
- The NA-ACCORD Steering Committee
- 3. Requesting NA-ACCORD data for approved CSs
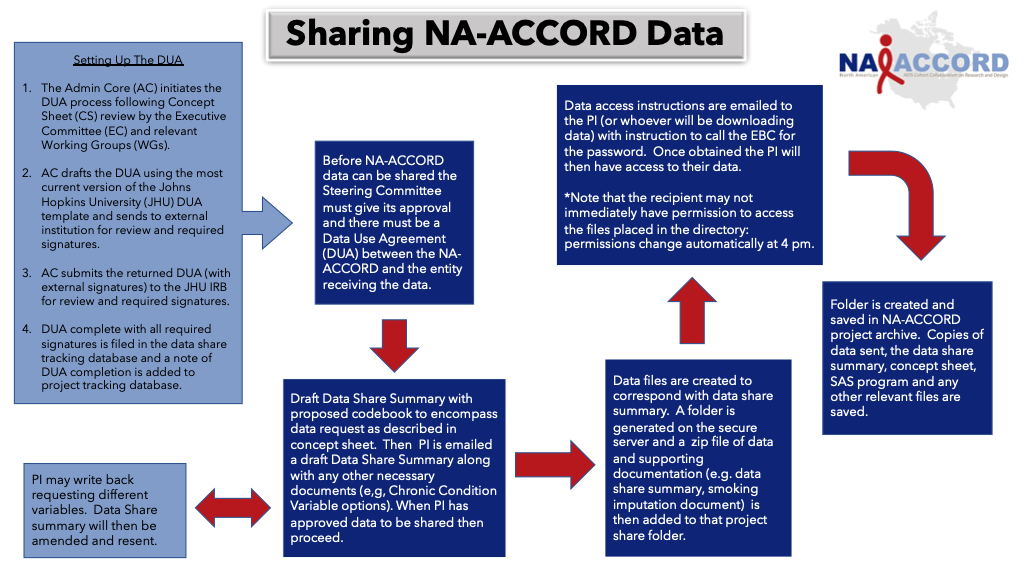
NA-ACCORD investigators who are interested in participating in your research will volunteer to join your Writing Group. You will utilize your writing group as potential co-authors for the scientific product that results from your research. The Writing Group is a valuable asset that allows access to a number of established HIV investigators in the US and Canada.
- 4. Abstract Approvals Process
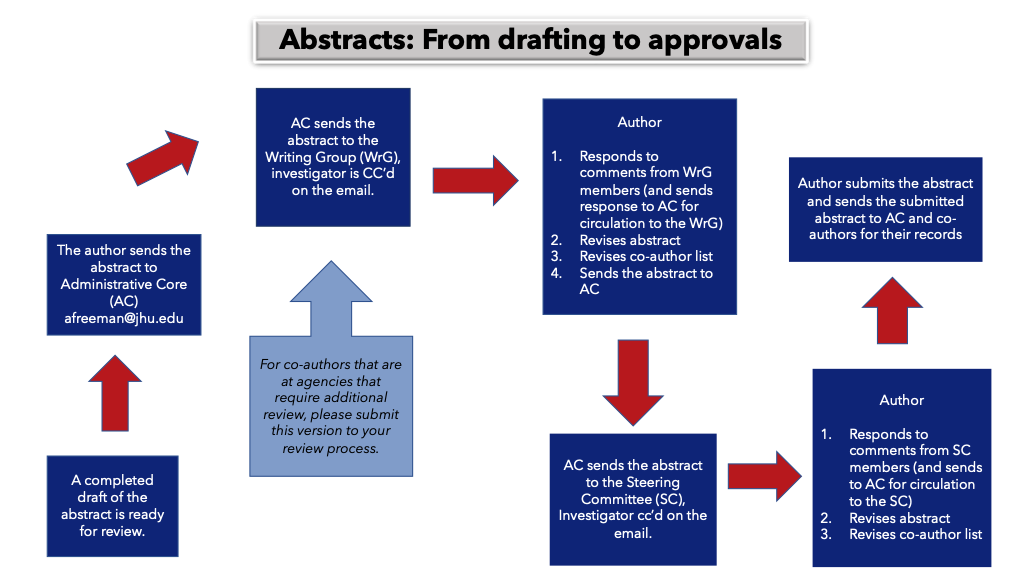
- 5. Manuscript Approvals Process
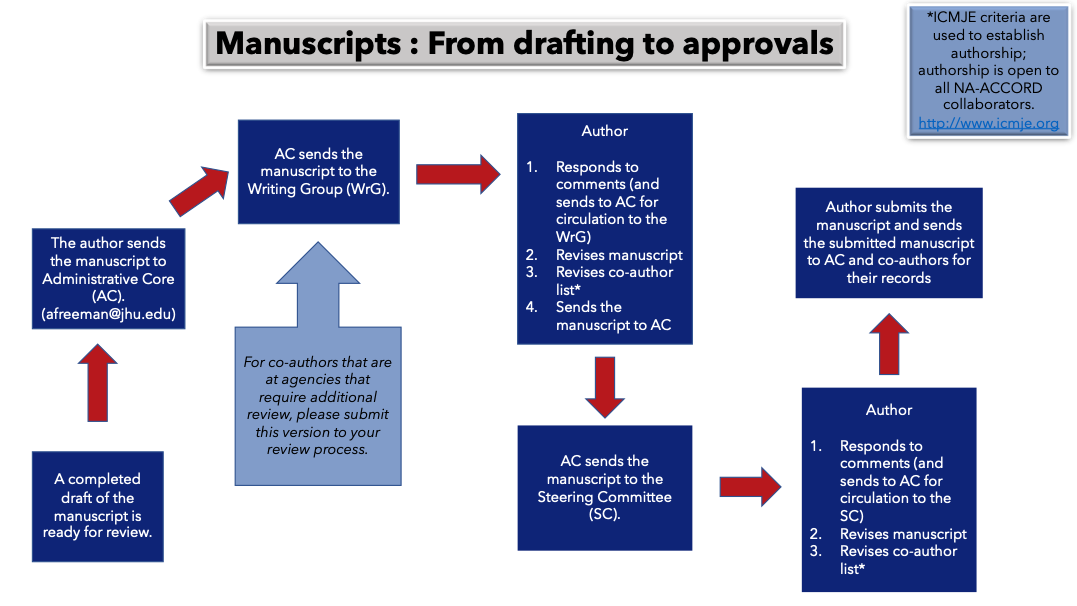
Once completed, your scientific product will be reviewed by the Steering Committee prior to making it public. If the product is a manuscript, please follow the Manuscript Checklist. Assistance is provided for completion of the Acknowledgements section.
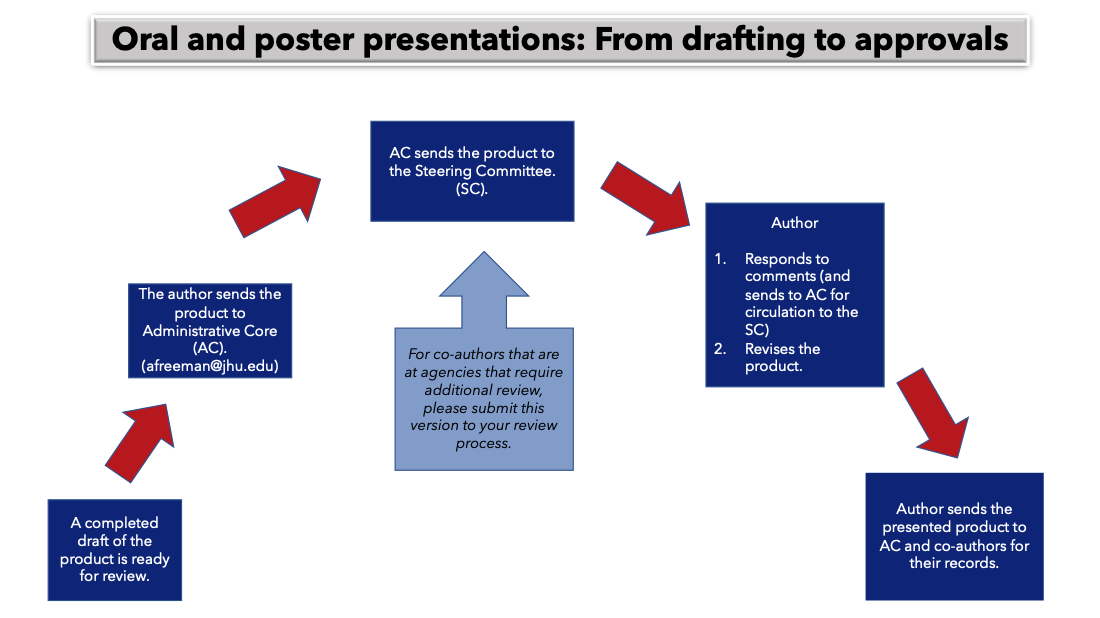
- 6. Oral and Poster Presentations Approvals Process
Files
| NAACCORD_Manuscript_Checklist_2014_0217.pdf |
| NA-ACCORD_proposal_development_process_flow_diagram.pdf |
| ConceptSheetTemplate_20200427.docx |